Cattle – Single-Dose Therapy: Enroflox® 100 is indicated for the treatment of Bovine Respiratory Disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida, Histophilus somni and Mycoplasma bovis in beef and non-lactating dairy cattle; and for the control of BRD in beef and non-lactating dairy cattle at high risk of developing BRD associated with M. haemolytica, P. multocida, H. somni and M. bovis.
Cattle – Multi-Day Therapy: Enroflox® 100 is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida and Histophilus somni in beef and non-lactating dairy cattle.
Swine: Enroflox® 100 is indicated for the treatment and control of swine respiratory disease (SRD) associated with Actinobacillus pleuropneumoniae, Pasteurella multocida, Haemophilus parasuis, Streptococcus suis, Bordetella bronchiseptica and Mycoplasma hyopneumoniae. Enroflox® 100 is indicated for the control of colibacillosis in groups or pens of weaned pigs where colibacillosis associated with Escherichia coli has been diagnosed.
Effective, ready to use and easy to inject
FDA approved
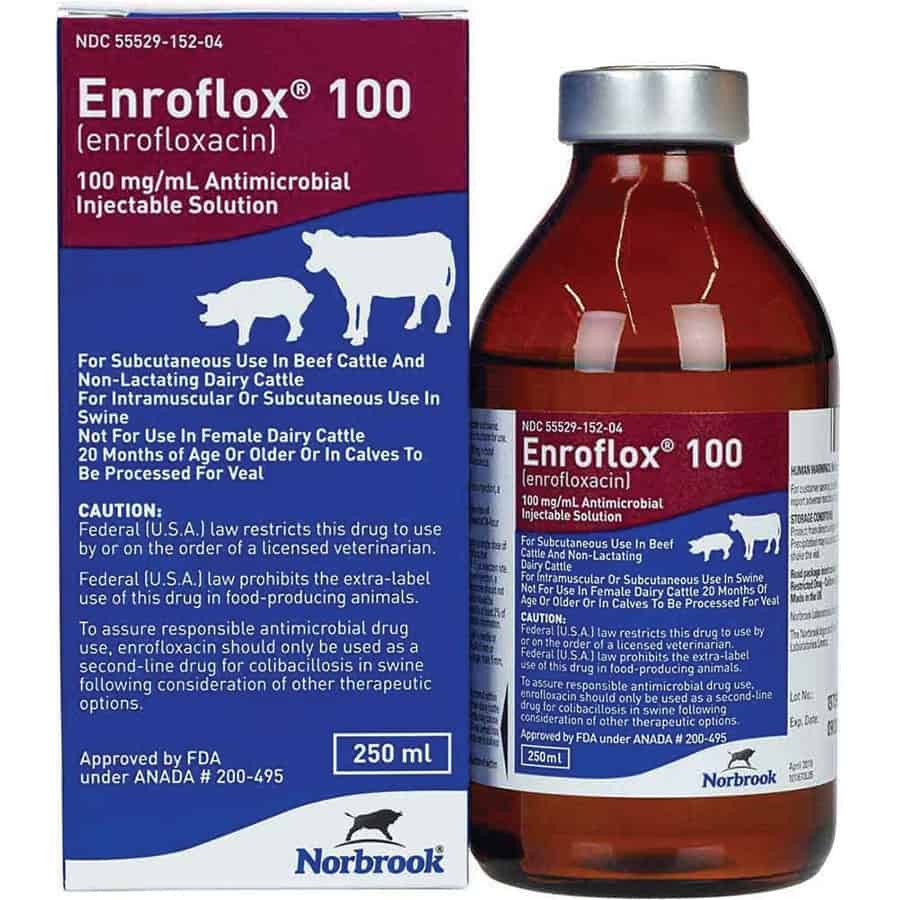
CAUTION:
Federal (U.S.A.) law restricts this drug to use by or on the order of a licensed veterinarian. Federal (U.S.A.) law prohibits the extra-label use of this drug in food-producing animals. To assure responsible antimicrobial drug use, enrofloxacin should only be used as a second-line drug for colibacilosis in swine following consideration of other therapeutic options.
Cattle intended for human consumption must not be slaughtered within 28 days from the last treatment. This product is not approved for female dairy cattle 20 months of age or older, including dry dairy cows. Use in these cattle may cause drug residues in milk and/or calves born to these cows. A withdrawal period has not been established in pre-ruminating calves. Do not use in calves to be processed for veal. To assure responsible antimicrobial drug use, enrofloxacin should only be used as a second-line drug for colibacillosis in swine following consideration of other therapeutic options. Swine intended for human consumption must not be slaughtered within 5 days of receiving a single-injection dose. Use with caution in animals with known or suspected CNS disorders. Observe label directions and withdrawal times. See product labeling for full product information.